 
The net ionic equation is a chemical equation which gives the ions that are participated in the formation of the final product. What is ionic and net ionic equations?Ĭomplete ionic equation is a chemical equation that explains the chemical reaction, clearly indicating the ionic species present in a solution. If the acid and base are equimolar, the pH of the resulting solution can be determined by considering the equilibrium reaction of HB⁺ with water. When a weak base and a strong acid are mixed, they react according to the following net-ionic equation: B(aq) + H₃O⁺(aq) → HB⁺(aq) + H₂O(l). 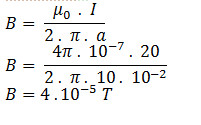
Which is a net ionic equation for the neutralization reaction of a strong acid with a weak base? The neutralization of a strong acid and strong base has a pH equal to 7. What is the reaction of acid base neutralization?Ī neutralization reaction is when an acid and a base react to form water and a salt and involves the combination of H+ ions and OH– ions to generate water. What is the net ionic equation for the reaction of any strong acid with a soluble strong hydroxide base such as NaOH group of answer choices?.What is the net ionic equation for the reaction of any strong acid with a soluble strong hydroxide base such as NaOH?.What is a neutralization reaction equation?.Which is a net ionic equation for the neutralization reaction of a strong acid with a weak base?. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
